|
|
 |
 |
|

|  |
Greetings!,
Below is a preview of Clearity's September 2010 Patient Newsletter. Click any title for the complete
article.
PARP Inhibitors - Fighting Cancer by Disabling DNA Repair
"Women with mutated BRCA can accumulate other DNA mutations that
eventually push cells down the path to cancer. Now scientists have found
a way to turn a BRCA mutation against the cancer cell."
Upcoming Events
Help Another Patient With A Donation
|
|
by Helen Gardner
|  | It had been a busy year, I was working as hard as ever, but I just didn't feel right. For the first time in 49 years, I had this little pooch in my stomach, not like any other pooch I had ever had. And it was just a little pooch. Everyone said, "Welcome to your 50's, welcome to menopause!" I went to the doctor several times in 2008 and in less than a year from my last gynecological exam, I was diagnosed with stage IIIC ovarian cancer. I had a rare type of ovarian cancer, clear cell. Finding answers and support for ovarian cancer would be challenging enough, now with a rare type of ovarian cancer, I would have to work even harder.
After my debulking surgery in my hometown of Dallas, Texas and 6 rounds of gold standard, first line treatment, my cancer appeared to be gone. But at my first 90 day check, the cancer was there. The news was devastating, and unfortunately, anticipated.
At the time of my recurrence, my oncologist in Dallas suggested that I enter a clinical trial. Alternatively, I could continue treatment with one of several second line, FDA approved drugs.
But how do I decide which trial would be the best? For that matter, how do I decide between a trial or second line treatment with FDA approved drugs, and even which FDA approved drugs? There are no guidelines with which to make a choice, other than the possible side effects of the chemo and data showing response rates to certain chemo treatments. I needed some way to evaluate the drugs that I could consider taking in my next step of this battle. I wanted a way to pick the drugs that would give me the best chance at eradicating my disease. I wanted to know if the drugs were going to work on MY cancer.
On New Year's Eve, while researching my options for clinical trials, I came across Clearity Foundation's information. My husband called fully expecting to get a recording and Laura, the founder of Clearity, answered the phone! From there she worked expeditiously to get samples of my tumor to her labs to begin testing. The molecular profiling testing Clearity Foundation offers could provide the answers to my questions, something to base my decision on. While waiting on the test results, I had appointments at Moffitt Cancer Center, a National Cancer Institute hospital in Tampa, Florida and in Dallas. I wanted to get many expert opinions for my next step.
What I learned in Dallas is that it is becoming more difficult to qualify for clinical trials as drug companies are adding criteria to their trials that exclude patients who have been exposed to multiple chemo regimens in the past. They are looking for "healthier" patients to test their drugs in an effort to get more accurate results for the drug's effectiveness. Also, there were limited trials in Dallas, one for solid tumors, one for vaccines, none for ovarian cancer specifically. This information furthered my insistence that I needed to find a suitable clinical trial. Waiting until after further treatments could exclude me from clinical trial options.
There were several ovarian cancer trials ongoing at Moffitt Cancer Center. One offered the trial drug in combination with a commonly used FDA approved drug. As my doctor in Tampa explained, I would be getting standard of care, PLUS a trial drug. After discussing the trial in Tampa with my doctor there, and reviewing my test reports with Clearity Foundation, I saw that trial as a true opportunity for me. Today, I have been on the trial for 8 months with plans to continue as I have experienced stable disease. This is better than what I believe I could have achieved on the second line treatment alone.
As Laura says, "The cure is out there," and I think that cure is specific to each person. We all have to keep trying until we find what will work for each of us. I am optimistic it will happen and I will keep trying until it does!
|
 | PARP Inhibitors: Fighting Cancer by Disabling DNA Repair |
|
by Cory Bentley, Ph.D
|  | 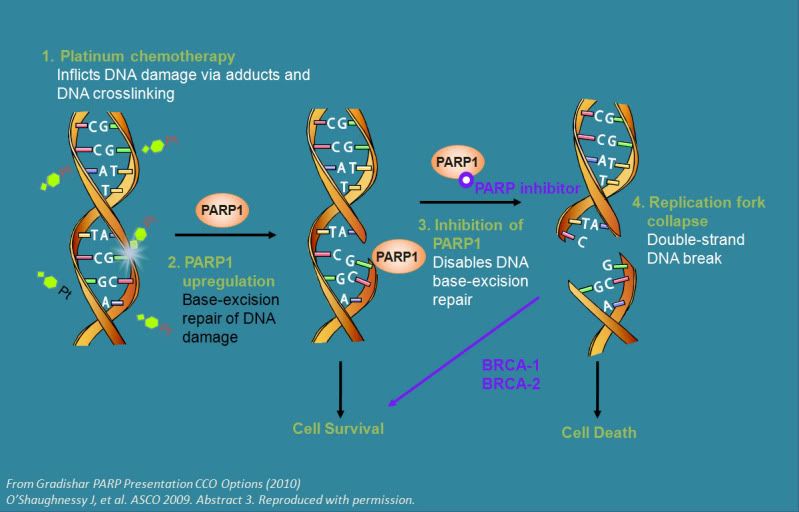 BRCA mutations are usually inherited and can lead to breast or ovarian cancer. BRCA (pronounced BRACK-uh) proteins are involved in DNA repair--the process that maintains DNA to keep it free from damage and to fix mutations as they happen. Women with mutated BRCA can accumulate other DNA mutations that eventually push cells down the path to cancer. Now scientists have found a way to turn a BRCA mutation against the cancer cell. BRCA mutations are usually inherited and can lead to breast or ovarian cancer. BRCA (pronounced BRACK-uh) proteins are involved in DNA repair--the process that maintains DNA to keep it free from damage and to fix mutations as they happen. Women with mutated BRCA can accumulate other DNA mutations that eventually push cells down the path to cancer. Now scientists have found a way to turn a BRCA mutation against the cancer cell.
A cancer cell divides rapidly, making two cells. Each of the two new cells needs a complete copy of the original cell's DNA to survive. In making all of this DNA, sometimes errors are made, so the new cells need some DNA repair to survive in this fast-paced world of proliferation. In patients that have BRCA mutations in their tumors, when a second DNA repair factor, PARP, is taken out with a drug called a PARP inhibitor, the cells just break down, and can no longer survive. In contrast, normal cells have the ability to repair the DNA damage because the BRCA pathway is active in those cells. This is the rationale behind two ongoing clinical trails of PARP inhibitors for ovarian cancer patients with BRCA mutations; however, a handful of other PARP inhibitor trials are open to patients who do not necessarily have a BRCA mutation. Deborah Zajchowski, PhD, scientific director at Clearity explains, "What is so exciting about this field is that we have learned that PARP inhibitors also appear to be active in patients without hereditary ovarian cancer. This means that even a greater number of patients may benefit from this treatment once we determine a way to identify those patients." These non-BRCA focused PARP inhibitor trials combine the PARP inhibitor with standard chemotherapeutics such as carboplatin and gemcitabine. Carboplatin and gemcitabine cause DNA damage in different ways. So just when the cancer cells need DNA repair most in order to survive, they get hit with PARP inhibition and die.
Recent work from the laboratories of Alan Ashworth and Christopher Lord, both from the Institute of Cancer Research in London, indicate that PTEN (pronounced pea-ten) is another important factor for DNA repair. PTEN is commonly mutated or is present at reduced levels in many cancers. Again, the double whammy of PTEN mutation and PARP inhibition may create a severe DNA repair deficit that cancer cells cannot overcome.
Levels of PTEN are evaluated in the tumor samples of Clearity patients as part of the tumor "blueprint" (What's behind the blueprint?). Although the connection between PTEN and PARP inhibition is new and untested in patients, information from patients' tumor blueprints may eventually help us understand if the PTEN-PARP connection is important and will help patients find the best treatment.
DNA damage has long been a mainstay of therapies against cancer. The development of a new form of DNA repair inhibition with PARP inhibitors looks promising as a potential cancer treatment. Clearity will be watching the ongoing clinical trials carefully and keeping our patients up to date.
|
 | Upcoming Events  |
 |  |
 DavidBartonGym Kicks Off Month-Long Charitable Program In Honor of Diane
Barton in New York, Miami, Chicago, and Seattle in hopes to raise
$100,000 for ovarian cancer with the proceeds going to The Clearity
Foundation and Ovarian Cancer Research Fund. For more information click here. To sign up click here. DavidBartonGym Kicks Off Month-Long Charitable Program In Honor of Diane
Barton in New York, Miami, Chicago, and Seattle in hopes to raise
$100,000 for ovarian cancer with the proceeds going to The Clearity
Foundation and Ovarian Cancer Research Fund. For more information click here. To sign up click here.
|
 | Help Another Patient With A Donation |
 |  |

Click here to donate and help another patient get access to a blueprint.
|
 | Thank You To Our Sponsor |
|
|
|
|
|
|
|
|
|