|
Skin Cancer as a Second Malignancy
Approximately 15% of CLL patients will develop skin cancer as a secondary malignancy. CLL negatively affects the immune system, and based on some evidence, is associated with deficient DNA repair. This DNA damage may lead to a higher susceptibility of developing skin cancer. The skin cancer is often more aggressive in CLL patients compared with people
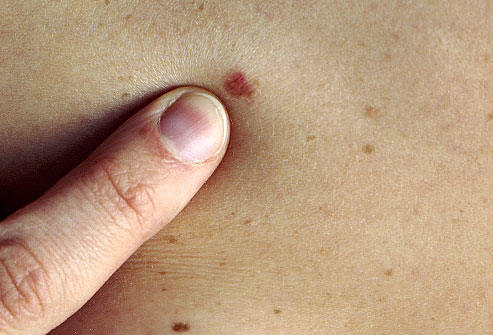 | |
Image provided by webmd.com
|
who have never had cancer, and CLL patients are at a higher
risk of developing recurrent skin cancer. Patients who need treatment for their CLL are at an even greater risk because their immune system is further compromised. Hopefully this paragraph has not increased your anxiety. Below we discuss what you can do to reduce your risk.
PROTECT YOUR SKIN. Avoiding the sun is ideal, but you cannot hide inside all of the time. Protective clothing is the best option for when you play outdoors. Sunscreen is important. Be smart about which one you choose and know what you are being protected from and what you are not. Wide brimmed hats are fashionable and functional. Sit in the shade when possible, and of course, avoid tanning and tanning booths.
BE ON THE LOOKOUT. Check for changes to your skin regularly. Here is a brief overview of what you should be looking for.
Squamous cell carcinoma is the most frequently reported skin cancer in CLL patients. Squamous cell can metastasize to other areas of the body. So if you see early signs, it is important to let your doctor know right away. Signs: rough, scaly patches that look like warts or open sores with a raised border.
Basal cell carcinoma is the most common skin cancer in general. It is commonly known as a rodent ulcer which burrows into the skin but does not spread. There are several different signs: A scar-like or irritated patch of skin that is white, yellow or reddish; a pink colored growth; a shiny, mole-like growth; an open sore that does not heal.
Merkel-cell carcinoma is very rare, as there are only 1,500 new cases in the US each year (less than 1% of the population). It is aggressive and tends to occur most frequently in people that are immune deficient. This means that there is an increased presence in the CLL population, although the odds are still very low. Signs: firm, reddish-pink nodule.
Melanoma is the most dangerous, but if it is detected early enough it is almost always curable. The problem is that melanoma often starts off looking like a regular mole. Know your moles and the ABCDEs of melanoma (as adapted from skincancer.org).
A is for Asymmetry. Noncancerous moles are usually symmetrical.
B is for Border. The border of moles should be clearly defined and even.
C is for Color. Moles with more than one color or an odd color should be looked at by a professional.
D is for Diameter. Larger moles, greater than 6 millimeters or ¼ inch are a warning sign.
E is for Evolution. Changes in size, shape, color, texture indicate something may be brewing.
TAKE CARE OF YOUR BODY. The immune system of CLL patients is already defunct. Keep yourself as healthy as possible by eating right and staying active. You will look and feel better too!
TALK TO YOUR DOCTOR. Keep your physician apprised of any changes or developments with your skin. Diligence is a good thing.
|