|
The NAHLN Quarterly |January 2011|
Volume 2, No. 4
THE OFFICIAL NEWSLETTER OF THE NATIONAL ANIMAL HEALTH LABORATORY NETWORK (NAHLN) A STATE AND FEDERAL PARTNERSHIP TO SAFEGUARD ANIMAL HEALTH |
|
Founding Principles and Features of the NAHLN | |
- Standardized, rapid diagnostic techniques
- Trained personnel, modern equipment
- Quality standards, proficiency testing
- Secure communication, alert reporting system
- Adequate facility biosafety/biosecurity levels
- Scenario testing
- Implemented quality management system
|
|
|
| |
For quick viewing, click below on the articles that interest you! |
|
Input Welcome! | |
We appreciate hearing from you! Are there other topics that you would like to hear about?
Please e-mail your comments to us at
|
|
Current Number of Subscribers: | |
1346
|
|
Upcoming Events | |
September 29-October 5, 2011:
54th Annual
American Association of Veterinary Laboratory Diagnosticians/
U.S. Animal Health Association Meeting
Buffalo, New York
|
|
|
|
Note from the Coordinator | |
I'm a person who sets goals and makes lists. Each December, I'm making more lists than usual as I set NAHLN Program goals for the following year. Keeping in mind the NAHLN mission and vision, as well as those of USDA, APHIS, Veterinary Services, and the National Veterinary Services Laboratories, I set goals based on the NAHLN operational and response gaps that were assessed during the previous year. This year's goals concentrate on the following topics:
- Improve emergency preparedness
- Increase visibility of NAHLN accomplishments
- Improve laboratory network operations
- Increase science focus
- Maintain and expand international collaboration
As I reviewed the relevant USDA projects to determine if our goals were in alignment, VS 2015 stood out as an effort quite familiar to those of you who have worked with us to develop and implement the NAHLN. VS 2015 is the strategic vision developed by leaders within Veterinary Services to guide the changes necessary to address animal health needs. Information on VS 2015 can be found on the USDA web site at: VS 2015. Below, I've copied a portion of one of the posted documents:
Q. How will stakeholders benefit from the VS 2015?
a. By emphasizing disease prevention, preparedness, detection and early response, VS hopes to minimize or avert industry-wide consequences. Better animal health capacities such as laboratory networks and veterinary stockpiles, will improve VS support when events do occur. In addition, creating greater flexibility will allow VS to shift personnel and funding more quickly to address emerging animal health issues. As a well-trained, practiced, nationally deployable response organization, VS can ensure that future responses throughout the United States will be enhanced. To support these efforts, VS plans to evaluate existing rules and regulations to ensure that they are science-based and meet the evolving needs of States and industry. In the past it was okay to take a one-size fits all regulatory approach, but in today's global economy we need greater flexibility to help States and industries maintain markets when disease strikes.
In conjunction with our State and Federal stakeholders, NAHLN has had the opportunity to put VS 2015 in action. We have worked together to increase our capabilities and capacities to detect and respond to adverse animal health events.
As I mentioned in the last edition of The NAHLN Quarterly, this edition includes the summary and recommendations of the 2010 Foot-and-Mouth Disease Table-top Exercises, and information on the capacity estimation program and the NAHLN Portal (see "Update on MTWG").
The NAHLN vision established at its inception continues to hold relevance and value. We have, and are continuing to facilitate the changes necessary to address evolving needs of States and industry.
Sincerely,

Barbara M. Martin
Coordinator
National Animal Health Laboratory Network
USDA, APHIS, VS, NVSL
|
|
|
|
| FMD Table-Top Exercises (TTX) Series Concludes | |
About the exercise series
The National Animal Health Laboratory Network (NAHLN) completed a three-part series of foot-and-mouth disease (FMD) exercises this past summer.
The first part of this series was a Veterinary Services' (VS) policy-level workshop that involved more than 25 personnel, including representatives from the National Center for Animal Health Emergency Management (NCAHEM), National Veterinary Services Laboratories (NVSL) - including the Foreign Animal Disease Diagnostic Laboratory (FADDL), National Surveillance Unit (NSU), National Veterinary Stockpile (NVS) Program, Swine Health Program, and the NAHLN Program Office. The primary goal of this workshop was to identify and discuss the roles and responsibilities of various decision-makers, as well as potential solutions to challenging policy questions related to NAHLN Laboratory response during an FMD outbreak, which would then be applied and further examined in the subsequent components of the NAHLN FMD exercise series.
The second component of the series was hosted by the National Agriculture Biosecurity Center (NABC) at Kansas State University (KSU). This table-top exercise, titled May Day II, focused on the actions, decisions, and communication of the NAHLN Laboratories, State Animal Health Officials and personnel, VS Area Office personnel and other units within VS, wildlife officials, and industry. Specifically, the objectives of this exercise were to examine early, mid, and late-response activities regarding the decision-making process for NAHLN activation and de-activation; testing capacity for the Kansas and Iowa State University Veterinary Diagnostic Laboratories in light of a theoretical FMD outbreak and surveillance sample collection protocols; testing algorithms after an FMD outbreak has been confirmed and for proving freedom from disease; and communication and coordination processes. Both Kansas and Iowa officials participated in this two-day table-top exercise.
|

| |
Participants examining maps of a simulated FMD outbreak scenario during the New England FMD Table-Top Exercise in Albany, New York
Photo courtesy of Claire Smith |
The third component of the series consisted of 15 separate table-top exercises, involving single States or multiple States in a region including one in which British Columbia, Canada participated. The objectives of these exercises were similar to the Kansas exercise; however, several of the lessons learned and resultant products from the Kansas exercise were applied in the subsequent exercises.
5 areas of preliminary findings
The following themes were consistent across the laboratories throughout the exercise series.
Laboratory Preparedness
Since the highly pathogenic avian influenza table-top exercise series in 2008, overall there is an increased understanding of the function and benefits of NAHLN. Additionally, NAHLN Laboratories have improved their preparedness for a foreign animal disease. However, it was reiterated that the laboratories need a set of disease outbreak guidelines that provide information regarding:
- Decision points for using biosafety level (BSL)-2 vs. BSL-3 spaces for completing different steps of the testing process
- Use and addition of proficiency tested (PT) personnel
- Compliance with Select Agent rule
- Timelines for receipt of reagents, support from other laboratories, and financial reimbursement
Communication
In general, there is effective and open communication among the State Animal Health Officials, VS' Area Offices and NAHLN Laboratories. The exercises reiterated the importance of early and frequent communication of outbreak events throughout the Network. Further, NAHLN Laboratories, State and APHIS field offices expressed the need from VS for education and information on the following:
- VS Memo 580.4
- National Veterinary Stockpile -- specifically the inclusion plan for FMD reagent supplies and timeline for activation
- Indemnity decision-making and timelines
- Surveillance and movement testing guidelines
- Wildlife policy
Laboratory Capacity
Consistently throughout this exercise series, we found that NAHLN Laboratories were well prepared for early outbreak testing capacity. The sustainability of that capacity, however, will be a challenge as the outbreak progresses. One of the unknown factors repeatedly raised was the long-term supply of diagnostic reagents. We also found that a real-time estimate of diagnostic capacity is vital for planning both in the laboratory and for the NAHLN Program both before and during an outbreak.
Diagnostic Development and Validation
There were a number of diagnostic development and validation needs expressed at almost every exercise. Those identified as important to be developed and deployable into the NAHLN Laboratories include:
- Validated test for FMD in milk
- Validation of pooled sample techniques
- Antibody ELISA
- DIVA antibody test capability
- Validation of tests in wildlife species (cervids, bison, elk)
Decision-Making
The majority of State Animal Health Officials and NAHLN Laboratories used VS Memo 580.4 as guidance in the decision-making process. The decisions that the States made about splitting suspect foreign animal disease samples between the NAHLN Laboratory and the Foreign Animal Disease Diagnostic Laboratory, as well as subsequent outbreak surveillance sampling strategies, greatly affects the testing volume, timing, and preparedness of the NAHLN Laboratory. States and laboratories also requested more information to improve their understanding on the VS decision-making processes.
Next Steps
Individual exercise reports will be shared with participants. An overall summary report, including findings and recommendations will be generated and shared with stakeholders.
Additional exercises will be held internal to the National Veterinary Services Laboratories to increase our internal preparedness with focus on decision-making, primary and support role activities, interaction and support of NAHLN Laboratories, collaboration, and communication.
A wrap-up VS policy workshop will be held in late spring 2011 to complete the policy matrix, which will include findings and any existing gaps in VS policy, from the first phase of the exercise series.
Article submitted by Sarah Tomlinson, Associate Coordinator, USDA/APHIS/VS/NVSL, NAHLN Program Office, Fort Collins, Colorado |
| Quality Assurance (QA) Update |
The American Association of Veterinary Laboratory Diagnosticians (AAVLD) Accreditation Committee has had a very busy year. At their most recent meeting they discussed site visit reports from 3 laboratories, responses to reports from 6 laboratories, and update reports from 8 laboratories. There are 8 laboratories on the agenda for site visits in 2011, one which will be visited for the first time. Auditor training for the Committee members and members of their audit pool is planned for 2011. The Accreditation Committee is also planning a symposium in conjunction with the Quality Assurance Committee and National Animal Health Laboratory Network at the Fall 2011 meetings to be held in Buffalo, New York.
A new question and answer forum is being planned for the Accreditation Committee to use to respond to questions regarding interpretation of the Requirements for an Accredited Veterinary Medical Diagnostic Laboratory in a timely manner. This will be available on the AAVLD web page. One question that auditors often hear is, "Why do we need to be accredited?" or "What does accreditation do for a laboratory?" There are many areas in which a laboratory may benefit upon achieving accreditation. Specifics for how each laboratory will benefit depend upon each laboratory's unique situation. Overall, accreditation provides certification of a laboratory's competence, which will increase confidence in the laboratory, provide additional funding opportunities, improve recognition of the laboratory, and provide continuous improvement of the laboratory as a whole. Attached are several examples of accreditation advantages:
Benefits of Accreditation
The increased efforts and expense involved in management and use of a quality system in order to achieve accreditation are often insignificant when compared to the advantages. As the laboratory processes and efficiency improve over time, the positive impact of accreditation far outweighs the perceived increased efforts made initially. These positive impacts are felt, not only in the laboratory that has earned accreditation, but in the profession overall.
Article submitted by Patricia Lukens, Quality Systems Manager, Washington Animal Disease Diagnostic Laboratory, Washington State University, Pullman, Washington |
| Getting to Know Us |
 | |
Jill Brown, Program Analyst,
NAHLN Program Office |
It is our pleasure to feature our very own Jill Brown for this edition's 'Getting to Know Us'. Jill joined the NAHLN Program Office in April of 2008. Jill wears many hats in our small program, including design and production of this newsletter - The NAHLN Quarterly. Jill's passion for marketing and her writing ability has also led to her involvement in generating other NAHLN reports and communication with both internal and external stakeholders. Another of Jill's primary functions for NAHLN is to report and monitor NAHLN's overall operational budget. A high priority for her is to always maintain fiscal responsibility and make the most with every Federal dollar. She states, "Through current budgetary constraints, the NAHLN Program continues to find innovative ways to move forward with our mission". She supports the NAHLN mission on a daily basis through maintenance and management of various funding mechanisms with the NAHLN Laboratories such as cooperative agreements and blanket purchase agreements. She maintains frequent communication with all of the NAHLN Laboratories, providing ever-friendly and responsive guidance through the Federal financial system.
Prior to joining NAHLN, Jill held administrative positions at the Center for Veterinary Biologics and the National Veterinary Services Laboratories' Diagnostic Virology Laboratory and the Program and Administrative Services group. Her 12 year Federal career in various units has provided her exposure and knowledge of both the veterinary diagnostic and veterinary biologics industries.
With an educational background in vocal performance, Jill spends a lot of her free time singing at various functions; a highlight for her was the privilege to sing the National Anthem in 2007 for the Secretary of Agriculture and his constituents during the dedication ceremony of the biolevel 3 animal agriculture containment facility of the National Centers for Animal Health. She also enjoys reading, running, bicycling, attending church functions, and traveling -- but most of all spending time with family and friends.
Jill is excited about the overall NAHLN concept and she quotes, "NAHLN is a common sense and logical next step toward protecting animal health - it benefits the U.S. financially, logistically, scientifically -- pooling our Nation's resources will better prepare us for an adverse animal health event".
We believe that Jill's dedication to effective customer service and fiscal responsibility with Federal funds will continue to make the difference to NAHLN and its successful partnerships and collaborations. Thank you Jill for all your hard work!
Article submitted by Sarah Tomlinson, Associate Coordinator, USDA/APHIS/VS/NVSL, NAHLN Program Office, Fort Collins, Colorado |
| Determining Diagnostic Testing Capacity in the NAHLN |
The Department of Homeland Security's (DHS) National Center for Foreign Animal and Zoonotic Disease Defense (FAZD) and the National Animal Health Laboratory Network (NAHLN) are collaborating to develop and implement a software program that is capable of capturing laboratory testing capacities in real-time. The NAHLN Capacity Estimation Program will increase the nation's capability to prepare and respond to a high consequence animal emerging and/or zoonotic disease by developing a software system to determine diagnostic testing capacity estimates in individual and across NAHLN Laboratories. The system and the data it generates will improve knowledge about individual and overall NAHLN diagnostic testing capacity, will aid in the modification of the NAHLN activation plan, and will assist in the prioritization of additional resources needed.
Needs - The NAHLN is tasked with surveillance, preparation, and response activities related to numerous high impact animal diseases by performing and reporting appropriate diagnostic tests. When a large-scale animal disease outbreak occurs, performing diagnostic tests on thousands of samples in an expeditious manner presents a major challenge. A coordinated systemic approach to optimize utilization of the NAHLN can be achieved by having a well conceived response plan and system to determine and monitor individual laboratory capacity. In order to be prepared to address animal disease outbreaks, the Animal and Plant Health Inspection Service (APHIS) must have a reliable and current estimate of diagnostic testing capacity in the NAHLN. Since laboratory-associated processes are frequently modified, a yearly assessment of diagnostic testing is necessary to determine the impact on the nation's overall diagnostic testing capacity. The development and implementation of a standardized capacity calculator would facilitate preparedness and also identify rate limiting steps in laboratory processes and assist in modification of the NAHLN activation plan.
 | |
Alfonso Clavijo and Gayne Fearneyhough, both from Texas Veterinary Medical Diagnostic Laboratory, discuss implications of a FMD outbreak
Photo courtesy of Stacy Morris |
Approach - The first phase of the research effort will begin with a capability assessment of NAHLN processes, equipment, capabilities, and staff resources. Laboratory personnel will work with NAHLN Program Staff and laboratories to assess the time effort associated with the performance of key laboratory tasks. A diverse set of existing laboratory capacity models, such as those used by other networks, will be analyzed for utility, and a subset selected for implementation into the capacity estimation program.
The second phase of the effort is the development and implementation of the capacity estimation program. The core of the program will be a secure web database application with a web browser front-end interface, enabling both centralized data storage/backup and remote privileged user access from anywhere. The schema will be tested and verified using sample data, and user acceptance testing will be conducted by 6 to 12 NAHLN Laboratories with a wide range of diagnostic testing capacities. Following verification and user testing, the prototype will be modified into a first generation capacity estimation program. It is expected that the web database application would be hosted on the secure National Center for Foreign Animal and Zoonotic Disease Defense (FAZD) web server. Upon acceptance of user testing, training materials will be finalized with input from NAHLN Program Staff and NAHLN Laboratories during the third phase of the research effort. Training will then be conducted with NAHLN laboratory personnel.
Outcomes - A web-based application to determine individual laboratory and an overall NAHLN capacity for diagnostic testing and scenario modeling of inputs related to constraints for all relevant laboratory activities including accessioning of samples, and all processes associated with tracking, testing, and storage of samples. The program will provide key metrics for "what if" scenarios, will predict laboratory capacity, and can be utilized to recommend a course of action based on which laboratory destinations and estimated result timeframes are identified. The design and implementation of the capacity calculator is such that it could easily be integrated as a "pluggable" dashboard component that is compatible with the Emergency Response Support System (ERSS) which is being developed under cooperative agreement for the APHIS Command Center in Riverdale, Maryland.
Customers - The application is to be developed specifically for the NAHLN as one tool to expand America's ability to detect, respond, and quickly recover from a high consequence animal emerging and/or zoonotic disease event. The application will be utilized at the individual state animal diagnostic laboratory level as well as at the national level for NAHLN Program Staff. The web-based application may also have future utilization for additional members of the Integrated Consortium of Laboratory Networks (ICLN) which includes such Federal agencies as the United States' Department of Agriculture, Department of Commerce, Department of Energy, Health and Human Services, Department of Interior, and Environmental Protection Agency.
Article submitted by Tammy Beckham, Laboratory Director, Texas Veterinary Medical Diagnostic Laboratory, Texas A&M System, College Station, Texas and Barbara Martin, Coordinator, USDA/APHIS/VS/NVSL, NAHLN Program Office, Ames, Iowa |
| NAHLN Surveillance Update | |
Wildlife Services' Wild Bird Avian Influenza (AI) Surveillance
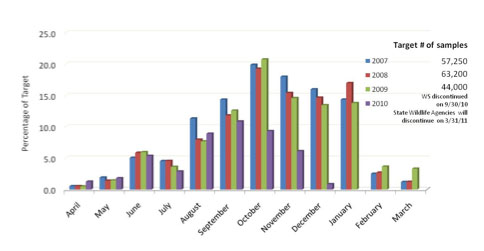
The fifth year of sampling wild birds for highly pathogenic avian influenza (HPAI) began April 1, 2010. Since then, wild bird sampling has served as an important early warning system for detecting HPAI in order to protect the poultry industry as well as to establish what low pathogenic avian influenza viruses are naturally circulating in the population. Samples are tested across 35 NAHLN Laboratories in States where the highest risk of HPAI entry via wild birds was determined based on a number of criteria deemed important by wildlife experts. With an anticipated discontinuation of funding, wild bird surveillance for HPAI will conclude on March 31, 2011.
 Bovine Spongiform Encephalopathy (BSE) Surveillance
Six (6) NAHLN Laboratories currently participate in enhanced BSE surveillance testing. BSE surveillance testing by NAHLN Laboratories began in June of 2004. The National Veterinary Services Laboratories' (NVSL) Pathobiology Laboratory in Ames, Iowa performs confirmatory testing.
The table below shows sample testing performed, by month, for BSE by NAHLN Laboratories (not including NVSL). The total number of samples tested for BSE by NAHLN Laboratories in the Federal fiscal year (FY) 2010 (October 2009 through September 2010) was 41,842 and 4,827 samples have been tested thus far in Federal FY 2011 (October and November 2010). Samples Tested for BSE by NAHLN Laboratories
FY 2010 and thus far in FY 2011
*BSE testing data provided by USDA/APHIS/Veterinary Services, National Surveillance Unit
Classical Swine Fever (CSF) Surveillance In January 2006, USDA implemented a CSF surveillance plan developed by the National Surveillance Unit in States (and Puerto Rico) with a high risk for introduction of CSF. Currently, there are 38 NAHLN Laboratories approved for CSF surveillance testing in addition to the NVSL's Diagnostic Virology Laboratory. The NVSL's Foreign Animal Disease Diagnostic Laboratory (FADDL) located in Plum Island, New York, performs confirmatory testing. Over 33,773 samples have been tested by NAHLN Laboratories since the inception of the program (not including NVSL). The tables below show the veterinary diagnostic laboratory (VDL) and slaughter sample testing performed, by month, for CSF surveillance by NAHLN Laboratories (not including NVSL). The total number of samples tested for CSF by NAHLN Laboratories in the Federal fiscal year (FY) 2010 (October 2009 through September 2010) was 6,294 (3,541 VDL/2,753 slaughter), and 916 samples (528 VDL/388 slaughter) have been tested thus far in Federal FY 2011 (October and November 2010).
VDL Samples Tested for CSF by NAHLN Laboratories
FY 2010 and thus far in FY 2011
___________________________________________
Slaughter Samples Tested for CSF by NAHLN Laboratories
FY 2010 and thus far in FY 2011
*CSF testing data provided by the USDA/APHIS/Veterinary Services, National Surveillance Unit
Chronic Wasting Disease (CWD) Surveillance Since 2001, the United States Department of Agriculture (USDA) has worked to develop and implement a CWD Program to prevent and control CWD in farmed/captive cervid herds and to conduct surveillance in free-ranging cervid populations to include deer, elk, and moose. The CWD Program includes a herd certification program that involves surveillance strategies to monitor for CWD in farmed/captive cervid herds, to respond to detections of CWD-positive cervids, and to investigate epidemiologically-linked animals. Interstate movement of cervids is also based on surveillance and herd certification status. Currently, there are 23 NAHLN Laboratories approved for CWD surveillance testing. The National Veterinary Services Laboratories' (NVSL) Pathobiology Laboratory in Ames, Iowa performs confirmatory testing. The table below shows the number of farmed/captive cervids tested, by month, for CWD by NAHLN Laboratories (not including NVSL). In Federal fiscal year (FY) 2010 (October 2009 through September 2010), 14,580 farmed/captive cervids were tested for CWD; an additional 5,419 cervids were tested by NVSL. Thus far in Federal FY 2011 (October and November 2010), 4,337 farmed/captive cervids have been tested for CWD; an additional 1,301 cervids were tested by NVSL.
Number of Animals Tested for CWD by NAHLN Laboratories
FY 2010 and thus far in FY 2011 *CWD testing data provided by the USDA/APHIS/Veterinary Services, National Veterinary Services Laboratories. Chart does not include 5,419 animals tested in FY 2010 and 1,301 in FY 2011 by NVSL. Scrapie Surveillance The National Scrapie Eradication Program is a USDA/APHIS/VS initiative with the objective of eradicating scrapie in U.S. sheep and goat populations. Efforts to eradicate scrapie have been ongoing since 1952; in 2001, an accelerated Scrapie Eradication Program was initiated. Since 2003, surveillance for the program has been conducted primarily through the Regulatory Scrapie Slaughter Surveillance (RSSS) component. Other surveillance activities include testing of scrapie-exposed and potentially exposed sheep and goats found through investigations of infected animals; clinical-suspect animals and other mature sheep and goats submitted veterinary diagnostic laboratories; rabies-suspect animals that test negative for rabies; and voluntary on-farm testing of flocks with risk factors for scrapie or as part of the Scrapie Flock Certification Program. Currently, there are 23 NAHLN Laboratories approved for scrapie surveillance testing. The National Veterinary Services Laboratories' (NVSL) Pathobiology Laboratory in Ames, Iowa performs confirmatory testing. The tables below show the number of animals submitted, by month, for scrapie testing by NAHLN Laboratories (not including NVSL). Table 1 shows the number of animals tested under the Regulatory Scrapie Slaughter Surveillance (RSSS) Program and Table 2 shows the number of animals tested for other scrapie surveillance (as described above). In Federal fiscal year (FY) 2010 (October 2009 through September 2010), 45,165 animals were tested under RSSS. 765 animals were tested for other surveillance (non-RSSS) in Federal FY 2010; an additional 1,232 animals were tested by NVSL.
Thus far in Federal FY 2011, 6,037 animals have been tested under RSSS. 463 animals have been tested for other surveillance (non-RSSS) thus far in Federal FY 2011; an additional 356 animals were tested by NVSL.
Number of Animals Tested for Scrapie by NAHLN Laboratories Regulatory Scrapie Slaughter Surveillance (RSSS) FY 2010 and thus far in FY 2011 ______________________________________________________
Number of Animals Tested for Scrapie by NAHLN Laboratories Other Surveillance (non-RSSS) FY 2010 and thus far in FY 2011 *Scrapie testing data provide by USDA/APHIS/VS, National Surveillance Unit and the NVSL. Chart does not include 1,232 animals tested in FY 2010 and 356 animals tested in FY 2011 by the NVSL.
VS' Swine Influenza Surveillance Veterinary Services has developed and implemented a surveillance plan for swine influenza virus (SIV), including an anonymous stream within participating NAHLN Laboratories. The addition of the anonymous option is designed to encourage pork producers to participate in SIV surveillance when their pigs show signs of respiratory disease. Within the anonymous stream, the results will only be identified back to the level of the State of origin, unless a producer requests in writing the traceable option. The SIV surveillance effort is designed to detect and identify circulating influenza viruses in swine. Additionally, this surveillance will provide epidemiological data related to genomic sequences. The objectives of SIV surveillance are to:
- Monitor genetic evolution of SIV to better understand endemic and emerging influenza virus ecology.
- Make SIV isolates available for research and to establish an objective database for genetic analysis of these isolates and related information.
- Select proper isolates for the development of relevant diagnostic reagents, updating diagnostic assays, and vaccine seed stock products.
Thirty-seven NAHLN Laboratories are testing swine samples for SIV surveillance. Since January 1, 2010 -- 2,158 swine samples (668 accessions) have been tested for SIV surveillance. The number of samples submitted for testing increased sharply in November 2010, when 490 samples from 174 accessions were submitted; prior to November, the monthly average of samples submitted for SIV testing each month was less than 200, derived from less than 50 accessions per month. Positive isolates are being further characterized (subtyping and sequencing) at NAHLN Laboratories, the National Veterinary Services Laboratories, Agricultural Research Service, and/or the Center for Disease Control to provide stakeholders with more complete information on State-level influenza virus ecology as the program further develops. Further results will be provided as they are completed. The surveillance information in this article has been provided by USDA/APHIS': Veterinary Services (VS)-National Center for Animal Health Programs, VS-National Surveillance Unit, VS-National Veterinary Services Laboratories (NVSL) and the NAHLN Program Office, Wildlife Services-National Wildlife Disease Program Charts in this article are based on information and test results available at the time of report generation. Numbers are subject to change due to later reporting of test results and updates in the database(s).
|
| NAHLN Laboratory Director Feature |
 | |
James Maxwell, NAHLN Laboratory Director
Bronson Animal Disease Diagnostic Laboratory
Kissimmee, Florida
Photo courtesy of Victor Alzona |
Meet Jim Maxwell, Director of two Florida laboratories - Bronson Animal Disease Diagnostic Laboratory, located in Kissimmee, Florida and Live Oak Animal Disease Diagnostic Laboratory located in Live Oak, Florida. He has served in this position since July 2008. Maxwell's career actually began as a member of the National Park Service Law Enforcement/Search and Rescue Rangers, where he performed wildfire duties in the western United States. Following a departure from this, he completed a zoology/chemistry pre-med Bachelor of Science degree at Northern Arizona University in Flagstaff, Arizona. He then earned his DVM at Colorado State University in Fort Collins, Colorado (an interesting side note - Barbara Powers, NAHLN Laboratory Director at Colorado State University was one of his instructors).
Maxwell entered into private practice for several years and in 2001 became a staff veterinarian at Live Oak Animal Disease Diagnostic Laboratory. In 2003, he was appointed Director of the Live Oak Laboratory, in 2007 was assigned to serve as Acting Director for the Bronson Laboratory, and in July 2008 he accepted the Laboratory Director position for both laboratories.
On the personal side, he enjoys fishing trips with family a couple of times each year.
Maxwell is quoted as saying the following regarding the NAHLN:
"I strongly believe NAHLN is important for our country. There will eventually be a large-scale animal disease outbreak for which NAHLN and NVSL Laboratories will collaborate to test large numbers of samples for the outbreak, provide proof-of-negative clearance testing after emergency response, and then provide continued surveillance results to ensure control measures and hopefully eradication of the disease agent. I believe NAHLN's organization, standardization, coordination, and support for animal disease laboratory operations will greatly enhance preparedness for protecting animal health. NAHLN truly is a beneficial State and Federal partnership and will hopefully gain more and more capability related to safeguarding human and environmental health as it relates to animal disease issues."
Article submitted by Stephanie Hadsall, Administrative Support Assistant, USDA/APHIS/VS/NVSL, NAHLN Program Office, Ames, Iowa |
| Information Technology (IT) Update |
Background. A critical aspect of the NAHLN is the effort to standardize data, improve data quality, and maximize the efficiency of data transfer via the IT infrastructure and data repository. The NAHLN IT system has been developed with data messaging (electronic, secure transfer of data) and standards to ensure accurate and consistent diagnostic information is quickly and securely transmitted.
Routine classical swine fever (CSF) test results have been electronically and securely submitted via a web-based system for over three years. In August 2008, NAHLN Laboratories began transmitting test results through standardized electronic messaging. Currently, there are 38 laboratories approved for CSF surveillance testing and 21 laboratories that are receiving surveillance samples. Thirteen laboratories are sending test results electronically.
Fifty-three NAHLN Laboratories are approved for avian influenza surveillance testing; thirty-five of avian influenza (AI)-approved laboratories are participating in Wildlife Services' (WS) Wild Bird AI Surveillance. The production system for the WS' Wild Bird AI Surveillance module was activated in June 2009. At present, twelve laboratories are actively messaging into the NAHLN IT system.
Update. Since October 2010, one additional laboratory has been approved to start messaging wild bird AI results. Currently, there are a total of four NAHLN Laboratories that have met the testing requirements to start production messaging AI results, with a start date pending. Additionally, messaging ELISA results for CSF from the one pilot laboratory performing that testing has successfully been underway since mid-October. Finally, the NAHLN IT team is working on the development of a swine influenza virus message, which is scheduled for implementation in early 2011. In 2011, emphasis will be placed on increasing the number of laboratories that message; additional funding for the enhancement of electronic messaging capability has been provided to 12 Member laboratories. Additionally, NAHLN will focus on developing and implementing processes to comply with the USDA's Information System security requirements.
Messaging Data. The graphs below show the percentage of results being electronically messaged for CSF and Wildlife Services' Wild Bird AI Surveillance programs. The remaining test results are being entered via the web-based Veterinary Services Laboratory Submissions (VSLS) database for CSF or are entered into the system for avian influenza by Wildlife Services' personnel. 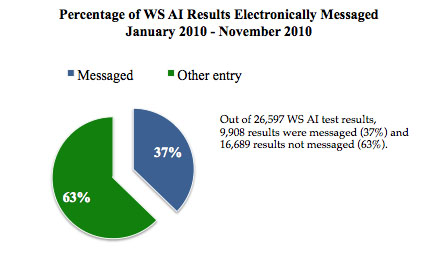 _____________________________________________________
*The messaging data has been provided by the APHIS, Veterinary Services, Office of the Chief Information Officer and testing provided by the APHIS, Veterinary Services, National Surveillance Unit. Charts are based on information and test/messaging results available at the time of report generation. Numbers are subject to change due to later reporting of test results and updates in the database(s). Article submitted by Leah Estberg, NAHLN Information System Subject Matter Expert, USDA/APHIS/VS, Office of the Chief Information Officer, Fort Collins, Colorado and Sarah Tomlinson, NAHLN Associate Coordinator, USDA/APHIS/VS/NVSL, NAHLN Program Office, Fort Collins, Colorado |
| Update on Methods Technical Working Group (MTWG) | |
Background: The National Animal Health Laboratory Network (NAHLN) Methods Technical Working Group (MTWG), Co-Chaired by Terry McElwain, Executive Director of the Washington Animal Disease Diagnostic Laboratory, and Barbara Martin, the NAHLN Coordinator is comprised of technical experts from NAHLN Laboratories, the National Veterinary Services Laboratories (NVSL), and other laboratories including representatives from USDA's Food Safety Inspection Service in Athens, Georgia, and the United States Geological Service/National Wildlife Health Center in Madison, Wisconsin.
The NAHLN MTWG functions as a vehicle for communication among NAHLN Laboratories on current methods, and provides input on various aspects of methods validation and approval of methods.
Update: The MTWG met this past December and were presented with an overview of the upcoming NAHLN Portal. The portal, a secure web site, has been built within the CoreSHIELD Framework, which was developed by multiple Federal partners with the purpose of supporting Federal, State and local governmental regulatory agencies and laboratories in defending the food supply through web-based tools that enhance threat prevention and response, risk management, communication and asset coordination, as well as public education.
The NAHLN Portal is being developed to provide preparedness data through a secure and collaborative web site. This approach promotes leveraging resources to produce products for multiple groups and networks.
Development of the following NAHLN Portal modules is expected to be completed in the next year:
- NAHLN Laboratory Directory - will contain information such as each laboratory's physical location, equipment, personnel listing, and information summarizing diagnostic capabilities.
- SOP Review and Release - will provide a mechanism for gathering stakeholder input into new and existing Standard Operating Procedures (SOPs) as well as a way to distribute SOPs specific to NAHLN.
- Validation training - will provide a tutorial on the various studies completed when validating an assay as well as definitions, reference materials, and examples of completed studies.
- Proficiency Testing - will be utilized for the announcement of proficiency test (PT) events, for requesting participation, submitting test results and receiving PT reports.
- Assay Performance Monitoring - will track the performance of assay controls and confidentially compare individual laboratory results with those of other laboratories. Alerts will be provided when controls are performing outside the expected range.
- Collaboration - will provide a collaborative site for document sharing and review, photo exhibits and galleries, video, discussion forums, calendar, online/offline meetings and more.
- Conference Management - will provide conference and meeting management capabilities for various activities such as announcing upcoming training or meetings, registration processes, and posting real-time training materials.
Members of the MTWG will have the opportunity to provide input and finalize the requirements for each module and participate in user acceptance testing. If you are interested in participating in the user acceptance testing process or have ideas to share, please contact the NAHLN Program Office by e-mail at nahln@aphis.usda.gov.
Article submitted by Barbara Martin, NAHLN Coordinator, USDA/APHIS/VS/NVSL, NAHLN Program Office, Ames, IA |
| NAHLN Procedure Manual References | |
Below you will find various NAHLN Procedure Manuals, the date of the latest version, and either a weblink or contact where they are available:
USDA/APHIS/Wildlife Services, Procedures Manual for Avian Influenza Surveillance, latest version April 2010, Contact: Tom DeLiberto at thomas.j.deliberto@aphis.usda.gov
Classical Swine Fever Surveillance Procedure Manual, latest version dated April 2007
Pseudorabies Surveillance Procedure Manual, Version 1.3, latest version dated October 2010
Procedure Manual for Vesicular Stomatitis Viruses (VSV), latest version dated January 2008
Influenza Surveillance in Swine Procedures Manual, latest version dated July 2010
|
|
| NAHLN Facts | |
The following links show a map and laboratory list of the laboratories that have been approved as part of the NAHLN testing network(including NVSL):
|
|
|
|
|