|
The NAHLN Quarterly |June 2009|
Volume 1, No. 2
THE OFFICIAL NEWSLETTER OF THE NATIONAL ANIMAL HEALTH LABORATORY NETWORK
A STATE AND FEDERAL PARTNERSHIP TO SAFEGUARD ANIMAL HEALTH |
|
Founding Principles and Features of the NAHLN |
|
- Standardized, rapid diagnostic techniques
- Trained personnel, modern equipment
- Quality standards, proficiency testing
- Secure communication, alert reporting system
- Adequate facility biosafety/biosecurity levels
- Scenario testing
| |
|
|
In This Issue |
Managing BSL 3 Labs
Follow-up on AI Tabletop Exercises
FADD Sampling Kits
WAVLD Pre-Meeting Symposium
Method OID Numbers
High Path AI in Wild Birds
Controlled Copy Process
Laboratory Designation
Upcoming Events
NAHLN Facts |
|
Following Up |
Update on the NAHLN Checklist
The revised NAHLN Checklist and Policy Document were sent to the NAHLN laboratories for completion by February 27, 2009. The completed checklists were reviewed by the NAHLN Program Office.
Laboratories were either fully or provisionally approved, based on accreditation status. Notifications and certificates were sent to the laboratories in April 2009. Site visits are currently being scheduled for provisionally approved laboratories.
NAHLN Program Staff |
|
Input Welcome! |
We appreciate hearing from you! Are there other topics that you would like to hear about?
Please e-mail your comments to us at NAHLN@aphis.usda.gov
We welcome your submissions.
If you would like to contribute an article, please contact us. | |
|
|
A Note from the Coordinator |
Welcome to the 2nd edition of the NAHLN Quarterly. The value of progress and partnerships has been obvious over the past several weeks as we've worked together to address concerns related to the 2009 novel H1N1 that has been isolated from humans. We greatly appreciate your willingness to participate in quickly organized calls and to provide information concerning testing capabilities. My personal thanks go out to the personnel from the Diagnostic Virology Laboratory, the National Animal Disease Center, the Southeast Poultry Research Laboratory, the Dossier Review Team, and the NAHLN Methods Technical Working Group. Our collaboration and coordination has made it possible for us to develop the diagnostic assays needed to detect the novel H1N1 as well as the processes necessary to approve, deploy, and assess performance of the assays. The lessons we've learned from our activities will be valuable as we continually improve our response capabilities and capacities.
Last year we focused our efforts on the NAHLN's ability to respond to adverse animal health events. We held table top exercises across the country and are in the process of releasing a summary report of those exercises; we collaborated with State and Federal animal health professionals to modify VS Memo 580.4 to include the potential use of NAHLN laboratories when testing samples from a foreign animal disease investigation; we developed and provided training for high-throughput testing; and we held a symposium in conjunction with the AAVLD meeting on emergency response. It was a busy and productive year!
This year our efforts are focused on documenting the processes necessary to ensure efficient and effective operation of the NAHLN. Here's a list of some of the processes we're working on:
-
Coordination of meetings and conference calls including minutes, and tracking action items;
-
Development, approval, and assessment of accomplishments associated with cooperative agreements and blanket purchase agreements;
-
Prioritization and coordination of information technology needs including development, deployment, and assessment of performance;
-
Laboratory review and assessment;
-
Training, including our "Train the Trainer" program;
-
Coordination of the proficiency testing process and reports;
-
Assay development, deployment and continual performance monitoring;
-
Development, delivery, and assessment of laboratory exercises
Please contact us if you are interested in having input into any of these processes.
I'm happy to report that our newsletter is now being distributed to over 400 individuals. If you know of others that are interested in our newsletter, feel free to forward this edition or have them contact the NAHLN Program office at NAHLN@aphis.usda.gov. We have had a few technical problems including the fact that the links in the 1st edition were live for a limited amount of time.
The NAHLN Program staff is determining how to ensure that we can archive the newsletter links so that they are accessed easily on our NAHLN website (NAHLN).
If you have suggestions on articles that should be included or would like to share an article, please contact us at NAHLN@aphis.usda.gov.
The information in our newsletter will also be shared in more detail in an annual report.
Thanks again for your continued partnership. Our commitment to working together is directly responsible for the NAHLN's success.
Sincerely,
Barbara M. Martin
NAHLN Coordinator
National Veterinary Services Laboratories
USDA, APHIS, VS | |
|
|
|
Advanced Topics in Managing BSL 3 Laboratories |
|
The Centers for Disease Control and the Eagleson Institute conducted a seminar "Advanced Topics in Managing BSL 3 Laboratories" January 27-28, 2009.
Topics addressed in the symposium included bio-risk assessment, occupational safety and health, public relations, laboratory emergencies, and animal model research issues.
The seminar, primarily targeted at research institutions, included items of note for animal disease diagnostic laboratories: the opinion that all high pathogenic avian influenza viruses, whether known to affect humans or not, should be handled in BSL 3 environments until inactivated; more universally acceptable standards for risk assessment are being developed; and, laboratories that work with high impact disease organisms or Select Agents should institute public relation programs to avoid the "not in my backyard" mentality.
The Eagleson Institute is a non-profit foundation whose mission is to promote the principles and practices of laboratory safety through seminars, training DVD's, software, scholarships and lectures. For a comprehensive list of resources and training seminars see www.eagleson.org or call (207) 490-1076.
The Eagleson Institute is currently planning the January 23-27, 2010 CDC International Biosafety Symposium, which will focus on safety in BLS3 and ABSL3 laboratories.
Article submitted by Wade Northington, Director, Associate Professor,
Breathitt Veterinary Center, Hopkinsville, KY |
| IT Update |
The NAHLN IT system is currently able to support reporting standardized classical swine fever (CSF) laboratory results electronically. The meaning and format of the result data transmitted are based on Health Level Seven (HL7) standards that have been harmonized with NAHLN laboratory, VS NAHLN program, and VS CSF animal health program information requirements. The vocabulary of the text-based information is standardized with LOINC, SNOMED, and HL7 terminology standards. The identifiers for key entities (e.g. specimen, laboratory, assay) are strengthened with the use of International Standards Organization globally unique identifiers (ANSI OID) and NAHLN program identifier quality standards (e.g. specimen barcode, assay specification). All standards are applied to improve the quality of the data exchanged. High data quality is anticipated to reduce the cost associated with linking unidentifiable entities and correcting errors sometimes introduced with manual data entry. It is also anticipated to improve the efficiency in use of information that is currently manually aggregated and grouped and open up new ways to utilize the information to support decision making.
Currently there are four laboratories successfully messaging CSF laboratory results. These results are stored in the NAHLN data repository and forwarded for translation and linking to CSF laboratory submission data for surveillance purposes.
In the near future, the NAHLN IT system will support reporting results for avian influenza (AI) testing of wild birds for Wildlife Services (WS). The near future plans also include providing support for sharing NAHLN documentation such as SOPs, laboratory emergency response plans, and exercise after action reports.
Article submitted by Leah Estberg, National Animal Health Laboratory Network IS Subject Matter Expert, USDA, APHIS, VS, Office of the CIO, Ft. Collins, CO.
|
| Request for Participants to Address Recommendations from A Wing and a Prayer" Summary and Analysis of the NAHLN HPAI Exercise Program |
As described in the first issue of the NAHLN Quarterly, the U.S. Department of Agriculture's Animal and Plant Health Inspection Service (APHIS) conducted 38 tabletop exercises in 2008 to test its emergency response capabilities in response to a potential outbreak of high-pathogenic avian influenza (HPAI) in the United States. Participating NAHLN laboratories sponsored the exercises from February to October in 2008. The exercises involved more than 700 participants representing 45 States and numerous Federal, State, and local agencies as well as the poultry industry.
After Action Reports were provided to participants for each exercise. A summary report was produced and has been used to identify gaps and prioritize necessary actions. APHIS has analyzed the summary report and outlined the recommendations. Working groups will be formed to prioritize necessary actions and develop plans to address gaps. Please contact the NAHLN Program Office at NAHLN@aphis.usda.gov if you are interested in participating in a working group to address laboratory issues.
The full report on the exercises will be released and posted on the USDA website. We will provide an e-mail update to our subscribers when the report is available.
Article submitted by Barbara M. Martin, Coordinator, USDA, APHIS, VS, NVSL, NAHLN Program Office, Ames, IA |
|
Preparedness |
|
Foreign Animal Disease Diagnostic (FADD) Sampling Kits
FADD sampling kits are supplied to field veterinarians, specifically foreign animal disease diagnosticians, to support their efforts for sampling animals which are showing signs consistent with potential foreign animal diseases. These kits supply most of the essential items needed by the veterinarian for collecting samples appropriate for rapid and accurate diagnosis. An FADD kit consists of two carrying cases which contain various supplies, and a shipping box for sending the samples to the NVSL laboratories for diagnosis. The two cases hold items such as safety glasses or goggles, a thermometer, Iowa hog holder, Yorkshire rope twitch, swine speculums, probangs of various sizes, hemostat, Dacron swabs, surgical blades with handle, syringes, needles of various gauges, vacutainer tubes for collecting blood, whirl pac bags, disposable gloves, a mesh glove, and a copy of the Merck Veterinary Manual. Various types of media in differing volumes are available from the NVSL such as Brain Heart Infusion media (BHI), Tris Buffered Tryptose Broth (TBTB), and Dulbecco's Modified Eagle Media (DMEM), to add to these kits to enable collection of tissue or swabs in media.

These kits are designed to be portable and self contained in order to make it easier for the
foreign animal disease diagnostician to collect the appropriate types and amounts of samples which will be needed for an accurate diagnosis to be made by the supporting laboratory.
Article submitted by Greg Mayr, Microbiologist, USDA, APHIS, VS, NVSL/FADDL, Plum Island, NY |
| WAVLD Pre-Meeting Symposium |
Development and Implementation of Veterinary Diagnostic Laboratory Networks - WAVLD Pre-Meeting Symposium
The NAHLN Program Staff has organized the Development and Implementation of Veterinary Diagnostic Laboratory Networks, The Principles of Laboratory Network Development and Function Pre-Meeting Symposium that will be held in conjunction with the 2009 World Association of Veterinary Laboratory Diagnosticians (WAVLD) meeting in Madrid, Spain on June 17, 2009. The topics discussed will include: Overview of the U.S. Laboratory Network (NAHLN); Quality Assurance and Accreditation; Assay Validation and Harmonization; Continuous Performance Assessment; Proficiency Testing / Ring Testing; Secure Communications and Reporting System; The Use of Modeling to Determine Laboratory Capacity; Scenario Testing; and Avian Influenza Wild Bird Surveillance in the U.S.
WAVLD registration and program information can be found at the following website: http://www.wavld2009.com/invitation.html. |
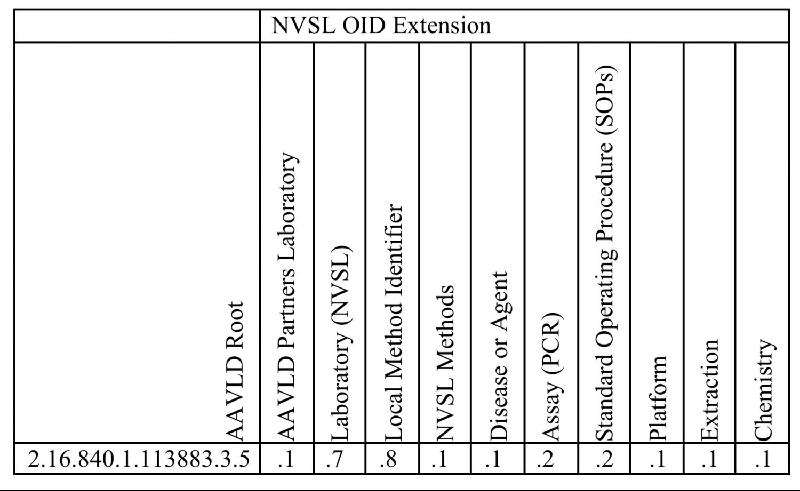
| Method OID Numbers |
The NAHLN Program staff is currently in the process of extending the Method OID numbers associated with validated assays to increase our understanding of the performance of assays and expand our ability to analyze test results data. Some commonly asked questions concerning OIDs and how they will be used and distributed follows.
What is a Method OID number?
OID stands for object identifier. The OID number is a unique number that represents an assay used in the laboratory information management system (LIMS) and electronic results messaging of the NAHLN laboratories.
Why would an extended Method OID number be important?
Extending the Method OID number allows analysis of data from NAHLN assays by identifying the SOP version as well as various parameters used to conduct a test such as amplification, extraction, and chemistry.
How will the additional information be used?
Having the additional information will give us the ability to monitor the performance of assays over time. If there are problems with a specific portion of an assay, for example, if we analyze the data and determine that one amplification platform isn't performing as well as another, we would be able to contact all the laboratories using that platform and explain the issues as well as potential solutions. The data could also be analyzed to determine if cutoffs need to be modified.
What does a NAHLN Method OID number look like?
A NAHLN Method OID is a series of digits separated by decimal point. The number between each decimal represents a specific value.
The breakdown of the additional data collected by the Method OID extension:

The Complete Map of a NAHLN Method OID number:
How will the new Method OID numbers be distributed?
The Method OID numbers will be distributed in an electronic spreadsheet as a matrix. The OID matrixes are being built by disease. Once a disease OID matrix is complete it will be sent to the NAHLN Laboratory Director and the NAHLN Laboratory IT Contact as designated by the Laboratory Director. As NVSL SOPs designated for NAHLN Laboratories are developed and finalized, they will be reviewed to determine if a NAHLN Method OID number needs to be developed or revised. The e-mail notification for the new Controlled Copy SOP(s) will include a revised OID Matrix and implementation deadlines. More information on the distribution of Controlled Copy and Uncontrolled Documents can also be found in this issue.
Who is involved in implementing Method OID numbers in NAHLN Laboratories? The NAHLN Laboratory IT Contact and NAHLN Laboratory personnel are expected to review the information and determine which OIDs apply to the methods being used in their laboratory. They are expected to make any necessary modifications to the LIMS and electronic results messaging. Prior OIDs will be archived in accordance to the individual NAHLN Laboratory's Quality System.
When will the new OID matrixes for current NAHLN assays be distributed?
The OID matrixes will be distributed by the NAHLN Program Office after they have been reviewed and finalized. Currently the OID Matrix for AI has been completed and reviewed and is in the process of being finalized. The CSF OID Matrix has been completed and is being reviewed. The FMD OID Matrix is in development. Since the OIDs for these assays were developed after the testing SOP was finalized and a controlled copy was provided to the NAHLN Laboratories, they will be provided as soon as they are complete. We anticipate that the OIDs will begin this spring and continue through the summer of 2009.
Article submitted by Traci J. Imlau, Program Assistant, USDA, APHIS, VS, NVSL, NAHLN Program Office, Ames, IA.
|
| Surveillance for High Pathogenicity AI in Wild, Migratory Birds* |
As the 2008 biological year for conducting high pathogenicity avian influenza (HPAI) surveillance in wild, migratory birds comes to an end, we wish to thank the state wildlife agencies, many Native American tribes, the NAHLN and NVSL, and Wildlife Services (WS) employees for their dedication and support in making the nationwide effort possible and a great success. Final results and accomplishments of the surveillance effort will be distributed in the next few months. Information provided below is a preliminary compilation of surveillance results and accomplishments.
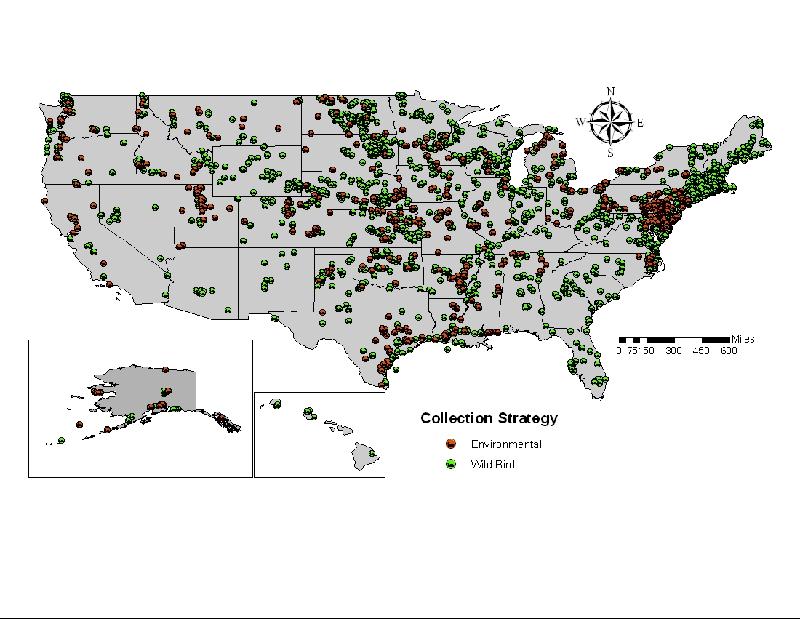
This year's efforts resulted in sampling of over 1,000 sick or dead birds, and over 63,000 apparently healthy birds (i.e., live bird, hunter harvested, sentinels). Similar to previous years, excellent sample distribution was achieved (Figure 1).
Figure 1.
 Many of the samples collected from morbidity/mortality events were submitted and diagnosed at U.S. Geological Survey (USGS) National Wildlife Health Center or NAHLN Laboratories. Over 26,000 fecal samples were collected in 31 states and submitted to WS National Wildlife Research Center for diagnostic testing. Most notable is that none of the wild bird or fecal samples have tested positive for HPAI. From the over 63,000 wild bird samples submitted to the NAHLN, almost 6,800 (10.8%) have tested positive for type A influenza by rRT-PCR. While these data are preliminary, we've noticed a marked increase in the percentage of samples testing positive this year.  Of the 6,800 positive samples, over 880 have screened positive for H5 or H7 when tested at NAHLN facilities. Fecal samples are pooled in batches of up to 5 samples prior to diagnostic testing, and approximately 150 pools have screened positive for the presence of type A influenza, with 3 of those pools testing positive for H5. Further screening at the NVSL determined that 20 samples from 13 States tested presumptive positive for both H5 and N1 by rRT-PCR. All of these have had viral sequences compatible with low pathogenicity avian influenza (LPAI) North American strains. Virus isolation results are pending for many of the samples, so virus isolation results and final rRT-PCR results will be published in the upcoming flyway reports. No HPAI has been detected in wild, migratory birds through this surveillance effort. Based on the enormity of the interagency surveillance effort, we are confident that our approach of balancing active, apparently healthy wild bird surveillance with morbidity and mortality investigations has yielded scientifically defendable data. Of the 6,800 positive samples, over 880 have screened positive for H5 or H7 when tested at NAHLN facilities. Fecal samples are pooled in batches of up to 5 samples prior to diagnostic testing, and approximately 150 pools have screened positive for the presence of type A influenza, with 3 of those pools testing positive for H5. Further screening at the NVSL determined that 20 samples from 13 States tested presumptive positive for both H5 and N1 by rRT-PCR. All of these have had viral sequences compatible with low pathogenicity avian influenza (LPAI) North American strains. Virus isolation results are pending for many of the samples, so virus isolation results and final rRT-PCR results will be published in the upcoming flyway reports. No HPAI has been detected in wild, migratory birds through this surveillance effort. Based on the enormity of the interagency surveillance effort, we are confident that our approach of balancing active, apparently healthy wild bird surveillance with morbidity and mortality investigations has yielded scientifically defendable data.
On behalf of the WS, National Wildlife Disease Program (NWDP), thank you for another successful year of surveillance. While we have been able to prove disease freedom of HPAI in wild, migratory birds, we have also learned a great deal regarding LPAI and determined many other causes of avian mortality.
*Data contained in this article are preliminary and should not be cited in other publications or print. For permission to cite this data, please contact the co-authors.
Article submitted by Seth R. Swafford, Assistant Wildlife Disease Coordinator and Kerri Pedersen, Disease Biologist, both from USDA, APHIS, WS, NWDP, Ft. Collins, CO. |
| Distribution of Controlled Copy and Uncontrolled Documents |
A process for distributing controlled copies of NAHLN related documents was implemented in 2007 to ensure that NAHLN Laboratories have the most recent version of applicable standard operating procedures (SOP). Questions concerning the process are answered below. Please let the NAHLN Program Office know if you have additional questions.
Which NVSL SOPs are distributed to the NAHLN Laboratories in a Controlled Copy format?
NVSL will identify all SOPs and forms that are used for assays conducted for NAHLN purposes. These along with other SOPs that impact NAHLN laboratories such as the SOP for corrective and preventative actions are identified as NAHLN documents in the NVSL document control system.
How are the NAHLN Laboratories notified that a Controlled Copy SOP is on the way?
An e-mail is sent to the NAHLN Laboratory Director, the Quality Manager, and Disease Contact Person as designated by the NAHLN Laboratory Director. The e-mail identifies the documents the NAHLN Laboratory will receive and includes electronic attachments such as form(s) that accompany the SOP and the Method OID matrix if necessary.
Who receives the Controlled Copy SOP?
The Controlled Copy SOP(s) are sent to the NAHLN Laboratory Director via Federal Express or any method where there is an acknowledgement of receipt. The NAHLN Laboratory Director documents that all Controlled Copy SOPs described in the cover letter/e-mail were received and distributed within their laboratory by completing the receipt form(s) and returning the receipt form(s) to the NAHLN Program Office. It is the responsibility of each NAHLN Laboratory to file and distribute the Controlled Copies in accordance with the individual NAHLN Laboratory's Quality System.
How can I tell if I'm looking at a Controlled Copy SOP?
A Controlled Copy Stamp will appear on the first page of the SOP. The NAHLN Program Office will initial, date and write in the Lab ID number when the document is prepared.
How can I tell a Controlled Copy SOP from a form? A Controlled Copy SOP will arrive in a hard copy format to be distributed in accordance to the NAHLN Laboratory Quality System. A form will be attached electronically to the e-mail notification and will be distributed in accordance to the NAHLN Laboratory Quality System. The type of document is identified in the document number:
SOP-NAHLN-1001.02 = SOP
FMA-NAHLN-1001.02 = Form A
OID-AIV-1200.01 = Method OID
Are the forms that are distributed electronically considered controlled copies?
No, the forms provided are not controlled copies.
Will NAHLN Method OIDs be distributed as described for Controlled Copy SOPs and forms?
The NAHLN Method OIDs will be distributed as an electronic spreadsheet matrix that is attached to the notification e-mail. The NAHLN Method OID matrix is the only electronic document provided in the e-mail notification that will have a receipt form included in the packet with the Controlled Copy SOPs.
For more information on when the initial NAHLN Method OID matrixes will be distributed please refer to the Method OID Numbers article in this issue.
Article submitted by Traci J. Imlau, Program Assistant, USDA, APHIS, VS, NAHLN Program Office, Ames, IA. |
| Laboratory Designation |
There are several levels of laboratory participation within the NAHLN. The term "core laboratories" was used to designate the original 12 participating laboratories. In 2007, the NAHLN Steering Committee created a system to define laboratory designations to reduce confusion among stakeholders. The laboratory designation system reflects different levels of infrastructure support for emergency-response preparation as well as funding for surveillance testing. Each level of laboratory participation is vital to the function and capacity of the NAHLN for early disease detection, surveillance, and surge-and-recovery testing in response to disease outbreaks.
How the NAHLN Laboratories Are Categorized
Four designations describe participation in the NAHLN: Core Member Laboratories and Member Laboratories (both of which receive infrastructure support), Contract Member Laboratories, and Adjunct Member Laboratories. The first three categories all participate in active surveillance (fee-for-testing) programs. Laboratories in all four categories have successfully completed the NAHLN approval checklist and quality-assurance requirements, and each has personnel trained and proficiency-tested to conduct the approved NAHLN diagnostic assays.
A Core Member Laboratory receives significant infrastructure support from, and also conducts fee-for-service testing for, the U.S. Department of Agriculture (USDA). This group of laboratories currently includes just the original 12. Their funding level enables them to be fully committed to the NAHLN mission and able to respond to domestic or foreign animal disease emergencies on a 24/7 basis.
A Member Laboratory receives limited annual infrastructure support from USDA for specific purposes, such as establishing information technology connections or developing capacity for data-reporting. These laboratories also conduct fee-for-service testing. Sixteen laboratories currently in this group could move to the Core Member category as additional funds become available to provide the significant annual infrastructure support needed to reach the Core Member category.
A Contract Member Laboratory performs only fee-for-service testing for control of specific animal diseases. These laboratories can move into either the Member or Core Member category as funding levels enable additional infrastructure support.
Because of its implementation of NAHLN protocols, an Adjunct Member Laboratory is considered a member of the NAHLN, but its primary mission is not domestic animal disease diagnostic work within the United States.
Over the past several months we have worked with the AAVLD/USAHA NAHLN Committee and the NAHLN laboratories to draft the responsibilities of each of the categories (see attached link: Laboratory Designation Table.pdf). NAHLN Program Staff is in the process of finalizing the requirements. If you have additional comments or suggestions, please provide them by June 19, 2009. The finalized requirements will be used to assess performance of individual laboratories.
Article submitted by USDA, APHIS, VS, NVSL, NAHLN Program Staff, Ames, IA. |
| Upcoming Events |
|
-XIV International Symposium of the World Association of Veterinary Laboratory Diagnosticians (WAVLD), Madrid, Spain, NAHLN Session, June 19, 2009. Link to: WAVLD Symposium.
-52nd Annual AAVLD/USAHA Meeting, October 7-14, 2009.
|
| NAHLN Facts |
|
The following links show a map and laboratory list of the laboratories that have been approved as part of the NAHLN testing network:
| |
|
|
|
|
|