Four Loko - Caffeine and DART

With the recent news about these caffeinated alcohol beverages, we decided to take a look and see what really is present. Thought most beverages have known amounts of caffeine, the popular Four Loko has been subject to wild speculation and rumors about its caffeine content. We used DART to get to the the bottom of this.
We used the automated sample preparation station we are developing, and spotted 4 replicates of each sample, with 13-C internal standards present. The samples were spotted onto the 3+D MicroWell screening plate and automatically analyzed with the 3+D Scanner and DART SVP source on an LCQ. We used a wide mass selection window to allow both the normal and labeled caffeine to pass simultaneously. And of course, no LC was used, just direct analysis in the open air.
We obtained good results that compare well with various reported values. As far as Four Loko - it has a lower concentration of caffeine than the other beverages, and, thus, is similar to a cup of coffee in terms of the amount per serving. However, people rarely have 4 cups of coffee in an hour, but that's another story.
A summary table is shown here where the red is the DART value and the blue is the reported value. Values on the left are in ppm.
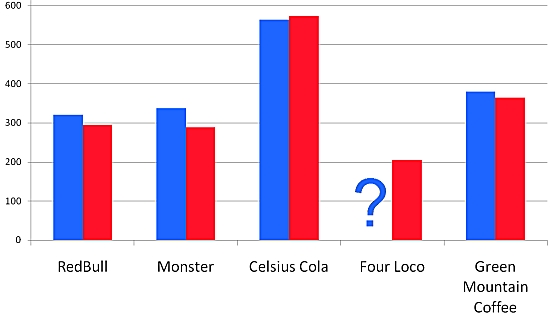 It is also interesting that the FDA is taking a hard look at these beverages and may regulate them in the future. According to their website, "the FDA has only approved caffeine as an additive for use in soft drinks in concentrations of no greater than 200 parts per million. It has not approved caffeine for use at any level in alcoholic beverages." For more on FDA's position and actions on this topic, you can look here. In the meantime the purveyors of these beverages will be submitting "Generally Regarded as Safe" (GRAS) studies so they can continue to sell their products. Should be interesting to watch.
If you would like more details on this analysis, please send me a note
|
DART Service Programs
 We have recently updated our service support to offer 1 year programs for the DART-SVP. It includes repair of all parts and labor. We pay for the shipping of the unit back to us where we repair it and send it back to you in a couple days. This should help you keep your system running in tip top shape. We have recently updated our service support to offer 1 year programs for the DART-SVP. It includes repair of all parts and labor. We pay for the shipping of the unit back to us where we repair it and send it back to you in a couple days. This should help you keep your system running in tip top shape.
If you would like details on the plans and pricing, just look here.
|
If you would like to keep up with the latest developments in the field of Open-Air Mass Spectrometry, please join here.

|
About IonSense
IonSense, Inc. provides open-air mass spectrometry solutions to the fields of food safety, forensics, drug development, and chemical analysis. They manufacture and develop direct analysis in real time (DART®) technology licensed from JEOL USA, Inc. and atmospheric solids analysis probe (ASAP™) licensed from M&M Consulting.
DART and ASAP Sources are available for most commercial LC/MS systems. Look here to see if your system is DART-ready. And check here to see if your system is ASAP-ready.
The latest news from IonSense:
IonSense Expands Product Line with ASAP Open-Air Ionization Source
|